Maharashtra SSC 10th Science Part 1 Exam 2026 : Most Important Questions Answers for Last-Minute Revision

The Maharashtra Board SSC 10th Science exam is on March 11, 2026, and this is your last chance to revise smartly. This last-day revision guide includes the most important questions with solutions:
We know how important it is to focus on the right topics and questions during revision, so this article provides information on the most likely Maharashtra Board SSC 10th Science questions for last-minute revision.
The questions above are taken from topics asked in previous year question papers. Check out their recurring pattern, which indicates their importance for the Maharashtra Board 10th Exam 2026.
Maharashtra Board Science Most Important Questions and Answers
Below are chapter-wise important questions and their concise answers for Maharashtra SSC Board Science Part 1 (Physics, Chemistry, Biology). These are based on recurring board patterns and key concepts.
Chapter 1: Gravitation
Q1. State Newton’s Universal Law of Gravitation.
Newton’s Universal Law of Gravitation states that every object in the universe attracts every other object with a force which is directly proportional to the product of their masses and inversely proportional to the square of the distance between their centers.
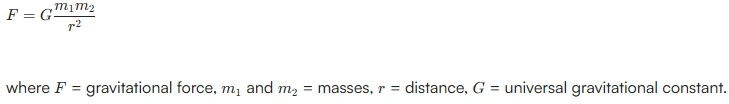
Q2. What is acceleration due to gravity? Write its value on Earth.
Acceleration due to gravity (g) is the acceleration produced in a body due to the gravitational force of the Earth. Its value on Earth is 9.8m/s2.
Chapter 2: Periodic Classification of Elements
Q1. State Mendeleev’s periodic law.
Mendeleev’s periodic law: The properties of elements are the periodic function of their atomic masses.
Q2. What is the modern periodic law?
Modern periodic law: The properties of elements are the periodic function of their atomic numbers.
Chapter 3: Chemical Reactions and Equations
Q1. Define a chemical reaction.
A chemical reaction is a process in which one or more substances (reactants) are converted into one or more new substances (products) with different properties.
Q2. Balance the following equation: Fe+H2O→Fe3O4+H2
Balanced equation: 3Fe+4H2O→Fe3O4+4H2
Chapter 4: Effects of Electric Current
Q1. State Ohm’s Law.
Ohm’s Law: The current passing through a conductor is directly proportional to the potential difference across its ends, provided the temperature remains constant.
Mathematically: V=IR
Q2. What is the SI unit of electric current?
The SI unit of electric current is ampere (A).
Chapter 5: Heat
Q1. Define specific heat capacity.
Specific heat capacity is the amount of heat required to raise the temperature of 1 kg of a substance by 1°C (or 1 K).
Q2. State the principle of calorimetry.
The principle of calorimetry: Heat lost by hot body = Heat gained by cold body (if no heat is lost to surroundings).
Chapter 6: Refraction of Light
Q1. State Snell’s Law.
Snell’s Law: The ratio of the sine of the angle of incidence to the sine of the angle of refraction is constant for a given pair of media.
Mathematically: sinrsini=constant
Q2. What is the refractive index of a medium?
Refractive index (n) is the ratio of the speed of light in vacuum to the speed of light in the medium.
Chapter 7: Lenses
Q1. State the lens formula.
Lens formula: f1=v1−u1 where f = focal length, v = image distance, u = object distance.
Q2. What is the power of a lens?
Power of a lens (P) is the reciprocal of its focal length in meters. P=f1 (in dioptres, D)
Chapter 8: Metallurgy
Q1. What is metallurgy?
Metallurgy is the process of extraction of metals from their ores and refining them for use.
Q2. Name two methods of refining metals.
- Electrolytic refining
- Distillation
Chapter 9: Carbon Compounds
Q1. What is homologous series?
A homologous series is a group of organic compounds having the same functional group and similar chemical properties, with successive members differing by −CH2− group.
Q2. Write the IUPAC name of CH3CH2OH.
IUPAC name: Ethanol
Chapter 10: Life Processes in Living Organisms
Q1. What is photosynthesis?
Photosynthesis is the process by which green plants prepare their own food using sunlight, carbon dioxide, and water, releasing oxygen as a byproduct.
Q2. Name the organelle where photosynthesis occurs.
Chloroplast
Chapter 11: Heredity and Evolution
Q1. What is heredity?
Heredity is the transmission of characteristics from parents to offspring through genes.
Q2. Who is known as the father of genetics?
Gregor Mendel
Chapter 12: Environment
Q1. What is ecosystem?
An ecosystem is a community of living organisms interacting with each other and with their non-living environment.
Q2. Name two greenhouse gases.
- Carbon dioxide (CO2)
- Methane (CH4)
SSC Maharashtra Board – Science Part 1: Important & Likely Questions (New Pattern)
1. Explain Newton's Universal Law of Gravitation with one real-life example.
2. How does weightlessness occur in space?
3. Derive the relationship between centripetal force and circular motion.
4. State Kepler's Three Laws of Planetary Motion with applications.
5. Explain Dobereiner's Triads and Modern Periodic Law.
6. How do atomic number and electronic configuration justify the long form periodic table?
7. Predict properties of elements based on periodic trends.
8. Explain the heating effect of electric current with diagram and application.
9. State and explain Fleming's Right Hand Rule and Left Hand Rule.
10. Why does a fuse wire melt earlier than a copper wire?
11. Write balanced equations for endothermic and exothermic reactions with examples.
12. Explain the role of catalysts with one example.
13. What is specific heat capacity? Explain its measurement.
14. Draw ray diagrams for refraction through a convex lens and derive lens formula.
15. How is focal length of a concave mirror determined experimentally?
16. Explain why stars twinkle but planets do not.
17. Describe extraction of iron from its ore.
18. Explain allotropy with reference to carbon.
19. Differentiate between saturated and unsaturated hydrocarbons.
20. Explain any one Indian space mission with objectives.
21. Explain the effect of gravitation on satellite motion.
---------------
👉 Also Check - Maharashtra Board Class 10 Study Material
Maharashtra Board Class 10 Study Material Get complete Maharashtra Board (MSBSHSE) Class 10 study resources including model papers, updated syllabus 2025-26, Balbharati textbooks, step-by-step textbook solutions, and quick revision notes for all subjects.
Everything you need to prepare smartly and score high in your SSC board exams – all in one place.







 Profile
Profile Signout
Signout












 Quiz
Quiz
 Get latest Exam Updates
Get latest Exam Updates 










